A) CaCl2 + Na2SO4 → CaSO4 + 2NaCl
B) KOH + HNO3 → H2O + KNO3
C) N2 + O2 → 2NO
D) AgNO3 + NaCl → AgCl + NaNO3
E) Al2(SO4) 3 + 6KOH → 2Al(OH) 3 + 3K2SO4
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
One mole of copper(II) sulfate,CuSO4 ,contains ________ moles of O.
A) 4
B) 6.02 × 1023
C) 1.51 × 1023
D) 2.41 × 1023
E) 2.41 × 1024
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
One mole of particles of any substance contains how many particles?
A) 106 particles
B) 3 × 10-10 particles
C) 3 × 1010 particles
D) 6.02 × 1023 particles
E) 6.02 × 10 - 23 particles
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Pentane ( C5H12 ) reacts with oxygen ( O2 ) to form carbon dioxide ( CO2 ) and water ( H2O) according to the following reaction.Answer the following question(s) about this reaction. C5H12 + ?O2 → ?CO2 + ?H2O -What is the coefficient for water in the balanced equation?
A) 2
B) 4
C) 5
D) 6
E) 8
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction of carbon with oxygen to produce carbon monoxide is an example of which class of reaction? 2C + O2 → 2CO
A) single replacement
B) double replacement
C) combination
D) catalytic
E) endothermic
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Barium chloride and sodium sulfate react according to the following equation. BaCl2 + Na2SO4 → BaSO4 + 2NaCl Answer the following question(s) about this reaction. -How many grams of barium chloride are needed to make 100.grams of barium sulfate?
A) 44.9 g
B) 89.2 g
C) 208 g
D) 233 g
E) 46.6 g
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
The moles of NH3 in 55.5 g of NH3 is ________.
Correct Answer

verified
Correct Answer
verified
True/False
The molar mass of chlorine gas is 35.45 g/mole.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In an oxidation-reduction reaction,the substance reduced always ________.
A) takes on oxygen atoms
B) loses electrons
C) gives up hydrogen atoms
D) gains electrons
E) becomes a charged species
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In an oxidation-reduction reaction,the substance oxidized always ________.
A) takes on oxygen atoms
B) loses electrons
C) gives up hydrogen atoms
D) gains electrons
E) becomes a charged species
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molar mass of copper(II) sulfate,CuSO4 ?
A) 16.00 g/mole
B) 63.60 g/mole
C) 111.6 g/mole
D) 159.6 g/mole
E) 319.2 g/mole
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The following reaction is endothermic.
2 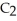
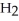 + 5
+ 5 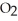 → 4C
→ 4C 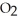 +
+ 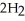 O + 310 kcal
O + 310 kcal
B) False
Correct Answer

verified
Correct Answer
verified
True/False
In the following reaction Al is oxidized.
2Al + 3 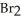 → 2
→ 2 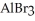
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Avogadro's number is the number of grams in a mole.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
0.400 mole of sucrose, C12H22O11 ,contains ________ C atoms.
A) 12
B) 6.02 × 1023
C) 5.02 × 1022
D) 2.89 × 1024
E) 4.80
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following question(s) ,consider the following equation. 2Mg + O2 → 2MgO -The number of moles of oxygen gas needed to react with 4.0 moles of Mg is ________.
A) 1.0 mole
B) 2.0 moles
C) 3.0 moles
D) 4.0 moles
E) 6.0 moles
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following question(s) ,consider the following balanced equation. Mg3N2+ 6H2O → 3Mg(OH) 2 + 2NH3 -How many grams of H2O are needed to produce 150 g of Mg(OH) 2 ?
A) 46 g
B) 18 g
C) 130 g
D) 93 g
E) 23 g
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following question(s) ,consider the following balanced equation. Mg3N2 + 6H2O → 3Mg(OH) 2 + 2NH3 -When 2 moles of Mg3N2 are allowed to react,how many moles of H2O also react?
A) 1 mole
B) 4 moles
C) 6 moles
D) 8 moles
E) 12 moles
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Pentane ( C5H12 ) reacts with oxygen ( O2 ) to form carbon dioxide ( CO2 ) and water ( H2O) according to the following reaction.Answer the following question(s) about this reaction. C5H12 + ?O2 → ?CO2 + ?H2O -In this reaction,what is the coefficient for calcium oxide? CaO + CO2 → CaCO3
A) 1
B) 2
C) 3
D) 4
E) 5
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
4.00 moles of sodium have a mass of ________.
A) 4.60 g
B) 11.0 g
C) 23.0 g
D) 44.0 g
E) 92.0 g
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 114
Related Exams