A) -2.18 × 10-19 J
B) +6.54 × 10-19 J
C) +4.58 × 10-19 J
D) -1.53 × 10-19 J
E) +3.76 × 10-19 J
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the velocity of a medicine ball (m = 10.0 kg) with a wavelength of 1.33 × 10-35 m.
A) 8.81 m/s
B) 12.3 m/s
C) 2.21 m/s
D) 4.98 m/s
E) 6.44 m/s
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the numbers for ml for an s orbital.
A) 0
B) -2, -1, 0, +1, +2
C) +1, +2, +3
D) 0, +1, +2, +3
E) +1
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many subshells are there in the shell with n = 2?
A) 1
B) 2
C) 3
D) 4
E) 25
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Give an example of a p orbital.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How much energy (in kJ) is required to ionize 2.52 moles of hydrogen atoms?
A) 1.65 × 103 kJ
B) 1.52 × 103 kJ
C) 5.49 × 103 kJ
D) 3.31 × 103 kJ
E) 5.89 × 103 kJ
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
________ is/are used to image bones and internal organs.
A) Ultraviolet light
B) Gamma rays
C) Microwaves
D) X-rays
E) Radio waves
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the energy of the green light emitted,per photon,by a mercury lamp with a frequency of 5.49 × 1014 Hz.
A) 2.75 × 10-19 J
B) 3.64 × 10-19 J
C) 5.46 × 10-19 J
D) 1.83 × 10-19 J
E) 4.68 × 10-19 J
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
During a thunderstorm,you see a flash of lightning.Five seconds later,you hear the thunder.How far away is the storm?
A) 1 yard
B) 10 miles
C) 1 mile
D) 6 miles
E) 12 miles
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the shortest frequency of light required to remove an electron from a sample of Ti metal if the binding energy of titanium is 3.14 × 103 kJ/mol.
A) 7.87 × 1015 Hz
B) 4.74 × 1015 Hz
C) 2.11 × 1015 Hz
D) 1.27 × 1015 Hz
E) 6.19 × 1015 Hz
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many photons are contained in a burst of yellow light (589 nm) from a sodium lamp that contains 623 kJ of energy?
A) 2.10 × 1013 photons
B) 3.06 × 1030 photons
C) 1.85 × 1024 photons
D) 4.03 × 1028 photons
E) 2.48 × 1025 photons
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the numbers for ml for a p orbital.
A) 0, +1
B) -1, 0, +1
C) -2, -1, 0
D) -2, -1, 0, +1, +2
E) +1, +2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Place the following types of electromagnetic radiation in order of decreasing energy. visible light radio waves infrared light
A) radio waves > infrared light > visible light
B) visible light > infrared light > radio waves
C) radio waves > visible light > infrared light
D) visible light > radio waves > infrared light
E) infrared light > radio waves > visible light
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the end (final) value of n in a hydrogen atom transition,if the electron starts in n = 1 and the atom absorbs a photon of light with an energy of 2.044 × 10-18 J.
A) 3
B) 4
C) 2
D) 5
E) 6
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of cycles that pass through a stationary point is called
A) wavelength.
B) amplitude.
C) frequency.
D) area.
E) median.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the energy of the orange light emitted,per photon,by a neon sign with a frequency of 4.89 × 1014 Hz.
A) 3.09 × 10-19 J
B) 6.14 × 10-19 J
C) 3.24 × 10-19 J
D) 1.63 × 10-19 J
E) 5.11 × 10-19 J
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When an electric current is passed through a tube containing neon,neon atoms absorb some of the electrical energy and reemit it as a ________ color.
A) white
B) yellow
C) violet
D) blue
E) red
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the energy change associated with the transition from 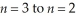 in the hydrogen atom.
in the hydrogen atom.
A) +3.03 × 10-19 J
B) -1.82 × 10-19 J
C) +5.51 × 10-19 J
D) -3.03 × 10-19 J
E) +2.69 × 10-19 J
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Each of the following sets of quantum numbers is supposed to specify an orbital.Choose the one set of quantum numbers that does NOT contain an error.
A) n = 4, l = 4, ml =-1
B) n = 3, l = 2, ml =+3
C) n = 4, l = 0, ml =-1
D) n = 3, l = 1, ml = -2
E) n = 5, l = 3, ml =-3
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following visible colors of light has the longest wavelength?
A) blue
B) green
C) yellow
D) red
E) violet
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 134
Related Exams