A) 12 amu
B) 16 amu
C) 18 amu
D) 32 amu
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is an endothermic reaction?
A) It is a reaction that requires heat as a reactant.
B) It is a reaction where the products have more energy than the reactants.
C) It is a reaction where there is a net adsorption of energy from a reaction.
D) all of the above
E) none of the above
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The chlorinated fluorocarbons molecules are acting as a(n) ________ by converting hundreds of thousands of molecules before they are inactivated.
A) catalyst
B) aerosol
C) active site
D) acid
E) base
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What are the formula masses of water, 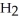 O; propene,
O; propene, 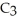
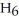 ; and 2-propanol,
; and 2-propanol, 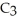
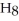 O?
O?
A) water: 18 amu; propene: 40 amu; 2-propanol: 58 amu
B) water: 18 amu; propene: 42 amu; 2-propanol: 62 amu
C) water: 18 amu; propene: 42 amu; 2-propanol: 60 amu
D) water: 18 amu; propene: 44 amu; 2-propanol: 64 amu
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following has the greatest mass?
A) 1 mole of Pb
B) 1 mole of H2
C) 1 mole of Be
D) 1 mole of Na
E) All have the same mass.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given that the above energy profiles have the same scale, which of the reactions would require the most energy?
A) a
B) b
C) c
D) d
E) none of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If it takes 20 beryllium atoms to equal the mass of two krypton atoms, what is the relative mass of beryllium compared to krypton?
A) 1/10
B) 1/20
C) 40 times
D) 100 times
E) 10 times
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How is Avogadro's number related to the numbers on the periodic table?
A) The atomic mass listed is the mass of Avogadro's number's worth of atoms.
B) The masses are all divisible by Avogadro's number, which gives you the weight of one mole.
C) The periodic table tells you the mass of one atom. From that, and Avogadro's number you know the number of moles.
D) The periodic table only gives us atomic numbers, not atomic mass.
E) The mass listed is Avogadro's number.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many grams of water, H2O, can be produced from the reaction of 25.0 grams of hydrogen, H2, and 225 grams of oxygen, O2?
A) 250 grams
B) 225 grams
C) 200 grams
D) 25 grams
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of H2 bonds are broken if you react nitrogen with hydrogen according to the following reaction? N2 + 3H2 → 2 NH3
A) 6
B) 2
C) 3
D) 1
E) 8
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following balanced equation, which has the highest coefficient? 4 H2 + 2 C → 2 CH4
A) H2
B) C
C) CH4
D) H4
E) none of the above
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which has more atoms: 64.058 g of sulfur dioxide, S 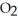 (64.058 amu) , or 72.922 g of hydrogen chloride, HCl (36.461 amu) ?
(64.058 amu) , or 72.922 g of hydrogen chloride, HCl (36.461 amu) ?
A) 64.058 g of S ![]() has more atoms than 72.922 g of HCl
has more atoms than 72.922 g of HCl
B) 72.922 g of HCl has more atoms than 64.058 g of S ![]()
C) 72.922 g of HCl and 64.058 g of S ![]() have about the same number atoms.
have about the same number atoms.
D) Not enough information is given.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following has the greatest number of particles?
A) 1 mole of Na
B) 22.990 g of Na
C) 1 mole of Be
D) 9.012 g of Be
E) All are the same.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If it takes three carbon atoms to equal the mass of one chlorine atom, what mass of chlorine do you need to equal the number of atoms in one kilogram of carbon?
A) 1/3 of a kg
B) 30 kg
C) 1 kg
D) 3 kg
E) 6 kg
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
You are given two samples of elements, and each sample has a mass of 10 grams. If the number of atoms in each of these samples is the same, what must be true of the two elements?
A) The density of the two elements are the same.
B) The elements are likely be located in the same position in the periodic table.
C) Their spectral patterns will likely be identical.
D) all of the above
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The atomic masses appearing in the periodic table are ________.
A) absolute masses
B) relative masses
C) standard deviation masses
D) formula masses
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Fourteen grams of nitrogen, N2, (N, atomic mass 14 amu) contains ________.
A) 1 mole of nitrogen atoms
B) 0.5 moles of nitrogen atoms
C) 0.25 moles of nitrogen atoms
D) Not enough information is given
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the number of moles of H2O produced if you combust 0.5 mole of CH4 according to the following balanced equation? CH4 + 2 O2 → CO2 + 2 H2O
A) 2 moles
B) 4 moles
C) 6 moles
D) 8 moles
E) 1 mole
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the environmentally unfriendly component of chlorinated fluorocarbons that ultimately damages ozone?
A) chlorine atoms
B) fluorine atoms
C) carbon atoms
D) molecular fragments of carbon and fluorine
E) all of the above
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the main difficulty in trying to eliminate chlorinated fluorocarbons from the atmosphere?
A) They are very stable and take a long time to decompose completely.
B) They are too light and blow away.
C) They do not absorb ultraviolet radiation.
D) all of the above
E) only a and b
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 150
Related Exams