A) rubidium perbromate
B) rubidium bromate
C) rubidium hypobromate
D) rubidium perbromite
E) rubidium bromide
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which species below is the nitrate ion?
A) NO2-
B) NH4+
C) NO3-
D) N3-
E) N3-
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
The possible oxidation numbers for iron are +1 and +2.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The gold foil experiment performed in Rutherford's lab __________.
A) confirmed the plum-pudding model of the atom
B) led to the discovery of the atomic nucleus
C) was the basis for Thomson's model of the atom
D) utilized the deflection of beta particles by gold foil
E) proved the law of multiple proportions
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An atom of the most common isotope of gold, 197Au, has __________ protons, __________ neutrons, and __________ electrons.
A) 197, 79, 118
B) 118, 79, 39
C) 79, 197, 197
D) 79, 118, 118
E) 79, 118, 79
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The correct formula for molybdenum(IV) hypochlorite is __________.
A) Mo(ClO3) 4
B) Mo(ClO) 4
C) Mo(ClO2) 4
D) Mo(ClO4) 4
E) MoCl4
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The charge on the copper ion in the salt CuO is __________.
A) +1
B) +2
C) +3
D) -1
E) -2
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The ions Ca2+and PO43- form a salt with the formula __________.
A) CaPO4
B) Ca2 (PO4) 3
C) Ca2PO4
D) Ca(PO4) 2
E) Ca3 (PO4) 2
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which atom has the largest number of neutrons?
A) phosphorus-30
B) chlorine-37
C) potassium-39
D) argon-40
E) calcium-40
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Of the choices below, which one is not an ionic compound?
A) PCl5
B) MoCl6
C) RbCl
D) PbCI2
E) NaCl
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Different isotopes of a particular element contain the same number of __________.
A) protons
B) neutrons
C) protons and neutrons
D) protons, neutrons, and electrons
E) subatomic particles
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Iron and chlorine form an ionic compound whose formula is FeCl3. The name of this compound is __________.
A) iron chlorine
B) iron (III) chloride
C) moniron trichloride
D) iron (III) trichloride
E) ferric trichloride
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Barium reacts with a polyatomic ion to form a compound with the general formula Ba3(X) 2. What would be the most likely formula for the compound formed between sodium and the polyatomic ion X?
A) NaX
B) Na2X
C) Na2X2
D) Na3X
E) Na3X2
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The name of the ionic compound NH4CN is __________.
A) nitrogen hydrogen cyanate
B) ammonium carbonitride
C) ammonium cyanide
D) ammonium hydrogen cyanate
E) cyanonitride
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Elements in Group 6A are known as the __________.
A) alkali metals
B) chalcogens
C) alkaline earth metals
D) halogens
E) noble gases
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Chromium and chlorine form an ionic compound whose formula is CrCl3. The name of this compound is __________.
A) chromium chlorine
B) chromium(III) chloride
C) monochromium trichloride
D) chromium(III) trichloride
E) chromic trichloride
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the empirical formula of the ionic compound that forms from aluminum and oxygen.
A) AlO
B) Al3O2
C) Al2O3
D) AlO2
E) Al2O
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which pair of elements would you expect to exhibit the greatest similarity in their physical and chemical properties?
A) As, Br
B) Mg, Al
C) I, At
D) Br, Kr
E) N,O
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The empirical formula of a compound with molecules containing 12 carbon atoms, 14 hydrogen atoms, and 6 oxygen atoms is __________.
A) C12H14O6
B) CHO
C) CH2O
D) C6 H7O3
E) C2H4O
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
What group in the periodic table would the fictitious element : 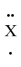 : be found?
: be found?
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 230
Related Exams