A) Why is calcium fluoride not used in toothpaste?
B) Why is calcium fluoride not used to fluoridate city water supplies?
C) Both A and B are good questions for the answer given.
D) Neither A nor B is a question that can be answered with the answer given.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solid has a solubility at room temperature of 78 grams per liter. If 1.0 L of a heated solution containing 100. g of solute is cooled to room temperature, how much solid is formed?
A) 22 g
B) 100 g
C) 78 g
D) 1 L
E) 78g/L
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A student is told to use 10.00 grams of sodium chloride to make an aqueous solution that has a concentration of 10.00 grams of sodium chloride per liter of solution. About how much water will she use in making this solution?
A) 10.08 L
B) 10.00 L
C) 9.992 L
E) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How is the solubility of a gas affected by temperature?
A) As temperature goes up, the solubility goes up.
B) As temperature goes down, the solubility goes down.
C) As temperature goes up, the solubility stays the same.
D) As temperature goes down, the solubility goes up.
E) both A and B
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements does not describes the similarity between soaps and detergents?
A) They have a polar ionic end and a long nonpolar tail.
B) Both dissolve oils.
C) Both are synthetic.
D) Both are biodegradable.
E) Both are made from fatty acids.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Many solvents expand to occupy greater volumes with increasing temperature. What happens to the concentration of a solution made with such a solvent as its temperature is increased?
A) Since concentration depends on how much mass is dissolved in a given volume, as the volume increases, the concentration decreases.
B) The concentration of a solution increases as the solute fits into the new spaces between the molecules.
C) Since it has a greater ability to dissolve more solute at a higher temperature, its concentration has decreased.
D) Since it has a greater ability to dissolve more solute at a higher temperature, its concentration has increased.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following accurately describes osmosis?
A) The more concentrated solution absorbs water from the less concentrated solution.
B) The less concentrated solution absorbs water from the more concentrated solution.
C) The less concentrated solution gets more dilute.
D) The ions migrate from the more concentrated solution to the less concentrated.
E) none of the above
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the strongest form of intermolecular attraction in a water molecule?
A) hydrogen bonding
B) induced dipole-induced dipole
C) covalent bonding
D) ion-dipole
E) polar-induced polar
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following intermolecular forces best describes why molecules like sucrose (which has many OH groups) are very water soluble?
A) dipole-dipole
B) induced dipole-induced dipole
C) dipole-induced dipole
D) ion-dipole
E) hydrogen bonding
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Does a plastic bottle of fresh water sink or float in the ocean? Why?
A) Sinks; The combined density of the plastic bottle plus the fresh water inside is greater than the density of the ocean water.
B) Floats; The bottle filled with fresh water floats in ocean water because it is less dense than the ocean water.
C) Floats then sinks; The bottle filled with fresh water floats in ocean water until the fresh water reaches the same temperature as the ocean water, at which point it sinks.
D) Sinks slightly; The added density of the plastic bottle will cause the bottle to sink slightly. It would most likely sink less than a foot where the density of the ocean water would be equal to or greater than the bottle of fresh water.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules is most likely to show a hydrogen bonding interaction?
A) CH3OH
B) CH3SH
C) CH4
D) H-C ![]() C-H
C-H
E) A, B and C
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would cost the least to purify by reverse osmosis?
A) agricultural runoff
B) sea water
C) brackish water
D) All of the above are the same.
E) None of the above can be purified by reverse osmosis.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Friends on a crowded ice skating rink tend to stay together. Strangers, however, tend to disperse. Is this behavior typical of atoms forming chemical bonds or molecules interacting? Explain.
A) No, atoms forming chemical bonds are just the opposite. Chemical bonds forming or molecules reacting would be more analogous to strangers tending to stay together and friends dispersing.
B) Yes, atoms forming chemical bonds are analogous to friends in the rink. Atoms held together by covalent bonds represent a molecule.
C) No, opposites (like strangers) attract to form stable covalent bonds in the world of atoms and molecules.
D) Yes and No; No, because this behavior is not typical of atoms forming chemical bonds, but yes since it is typical of molecules interacting.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Dipole-induced dipole forces of attraction exist between water and gasoline, and yet these two substances do not mix because water has such a strong attraction for itself. Which of the following compounds might best help to make these two substances mix into a single liquid phase? 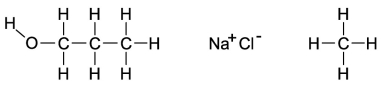
A) the molecule on the far left because the O-H bond is polar and the carbon and hydrogen bonds are nonpolar
B) the molecule in the middle because when the salts mix into the water, it will help separate the water and decrease the attraction for itself
C) The molecule on the right will form attractions with the polar ends of the water, allowing the gasoline a chance to mix with the water.
D) All of these molecules would be equally effective at increasing the mixing of gasoline and water.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is happening at the molecular level when a polar molecule like water interacts with a typical sodium ion?
A) The water molecule aligns such that the oxygen interacts with the sodium.
B) The water molecule aligns such that the hydrogens interact with the sodium.
C) The polarity of the water molecule is altered making the oxygen more positively charged.
D) The polarity of the water molecule is altered making the hydrogens more negatively charged.
E) none of the above
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How are intermolecular forces and solubility related?
A) Solubility depends on the solvent's ability to overcome the intermolecular forces in a solid.
B) Solubility depends on the solute's ability to overcome the intermolecular forces in the solvent.
C) Solubility is a measure of how strong a solvent's intermolecular forces are.
D) Solubility is a measure of how weak the intermolecular forces in the solute are.
E) none of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following describes the term concentration?
A) It is what you are doing now to answer this question.
B) It is the amount of solute in a given amount of solution.
C) It is the amount of solvent in a given amount of solution.
D) It is the given amount of solution in a given container.
E) It is the given amount of solvent per amount of solute.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why might softened water not be good for some one who is trying to reduce their dietary sodium ion intake?
A) The potassium used in the softener to replace the calcium and magnesium ions increases the sodium retention in the body.
B) Water softeners work by replacing the calcium and magnesium ions of the water with sodium ions; softened water contains increased levels of sodium ions.
C) The softened water leaches sodium from the body, so a person might not be getting the dietary recommended levels of sodium.
D) A person needs a certain level of calcium and magnesium in the water to help excrete the sodium from their body.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which has the most atoms?
A) a mole of gold
B) a mole of helium
C) a mole of lead
D) All of the above have the same number of atoms.
E) none of the above
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following solutions is the most dilute?
A) one liter of water with 1 gram of sugar
B) one liter of water with 2 grams of sugar
C) one liter of water with 5 grams of sugar
D) one liter of water with 10 grams of sugar
E) They all have the same volume.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 157
Related Exams