A) H2CO3
B) HCO3-
C) H2O
D) H3O+
E) CO2
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of a 0.014 M Ca(OH) 2 solution?
A) 1.85
B) 1.55
C) 12.15
D) 12.45
E) 15.85
G) A) and B)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
The equilibrium constant for the reaction C7H15COOH(aq) + HCOO-(aq) 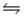 C7H15COO-(aq) + HCOOH(aq)
Is 7.23 × 10-2 at 25°C.If Ka for formic acid (HCOOH) is 1.77 × 10-4, what is the acid dissociation constant for C7H15COOH?
C7H15COO-(aq) + HCOOH(aq)
Is 7.23 × 10-2 at 25°C.If Ka for formic acid (HCOOH) is 1.77 × 10-4, what is the acid dissociation constant for C7H15COOH?
A) 2.45 × 10-3
B) 4.08 × 10-2
C) 7.81 × 10-4
D) 1.00 × 10-4
E) 1.28 × 10-5
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The hydronium ion and the hydroxide ion, in that order, are:
A) H3O+, OH+
B) OH-, H3O-
C) OH-, H+
D) H3O+, OH-
E) H3O-, OH-
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
Which of these acids is stronger, H2SO4 or HSO4-?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the pH of a KOH solution made by mixing 0.251 g KOH with enough water to make 1.0 × 102 mL of solution.
A) 1.35
B) 2.35
C) 7.00
D) 11.65
E) 12.65
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
The pH of a 0.6 M solution of a weak acid is 4.0.What percent of the acid has ionized?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following solutions is acidic?
A) [H3O+] = 7.0 x 10-7 M
B) [H3O+] = 1.5 x 10-10 M
C) [H3O+] < 7.0 x 10-7 M
D) [H3O+] > 7.0 x 10-7 M
E) [H3O+] = 1.0 x 10-14 M
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is not a conjugate acid-base pair?
A) H3PO4 and H2PO4-
B) H2PO4- and HPO42-
C) H3PO4 and HPO42-
D) HPO42- and PO43-
E) H2O and H3O+
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the concentration of oxalate ion (C2O42-) in a 0.175 M solution of oxalic acid (C2H2O4) .[For oxalic acid, Ka1 = 6.5 × 10-2, Ka2 = 6.1 × 10-5.]
A) 0.11 M
B) 6.1 × 10-5 M
C) 4.0 × 10-6 M
D) 0.0791 M
E) 0.175 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the reaction HSO4-(aq) + OH-(aq)  SO42-(aq) + H2O(l) , the conjugate acid-base pairs are
SO42-(aq) + H2O(l) , the conjugate acid-base pairs are 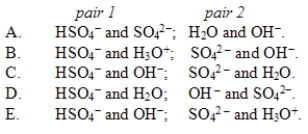
A) A
B) B
C) C
D) D
E) E
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hard water deposits (calcium carbonate) have built up around your bathroom sink.Which one of these substances would be most effective in dissolving the deposits?
A) ammonia
B) bleach (sodium hypochlorite)
C) lye (sodium hydroxide)
D) vinegar (acetic acid)
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
If the pH of liquid bleach is 12.0, what is the hydroxide ion concentration in this solution?
Correct Answer

verified
1 × 10-2 M
Correct Answer
verified
Multiple Choice
Which of these species will act as a Lewis acid?
A) NH3
B) NH4+
C) H2O
D) BF3
E) F-
G) A) and D)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
The pOH of a solution is 10.40.Calculate the hydrogen ion concentration in the solution.
A) 4.0 × 10-11 M
B) 3.6 M
C) 4.0 × 10-10 M
D) 2.5 × 10-4 M
E) 1.8 × 10-4 M
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of a solution prepared by mixing 50.0 mL of 0.300 M HCl with 450.0 mL of 0.400 M HIO3? [Ka(HIO3) = 1.6 × 10-1]
A) 1.52
B) 0.80
C) 0.72
D) 0.89
E) 0.66
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the H+ ion concentration in a 4.8 × 10-2 M KOH solution?
A) 4.8 × 10-2 M
B) 1.0 × 10-7 M
C) 4.8 × 10-11 M
D) 4.8 × 10-12 M
E) 2.1 × 10-13 M
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following yields a basic solution when dissolved in water? I.NH3 II.Na2O III.LiOH IV.P4O10
A) I and II
B) I and IV
C) II and III
D) I, II, and III
E) I, II, III, and IV
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The equilibrium constant for the reaction C6H5COOH(aq) + CH3COO-(aq)  C6H5COO-(aq) + CH3COOH(aq)
Is 3.6 at 25°C.If Ka for CH3COOH is 1.8 × 10-5, what is the acid dissociation constant for C6H5COOH?
C6H5COO-(aq) + CH3COOH(aq)
Is 3.6 at 25°C.If Ka for CH3COOH is 1.8 × 10-5, what is the acid dissociation constant for C6H5COOH?
A) 5.0 × 10-6
B) 6.5 × 10-5
C) 2.3 × 10-4
D) 8.3 × 10-5
E) 5.6 × 10-6
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the pH of a carbonated beverage in which the hydrogen ion concentration is 3.4 × 10-4 M.
A) 2.34
B) 3.47
C) 6.01
D) 7.99
E) 10.53
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 178
Related Exams