A) 0.
B) 1.
C) 2.
D) 3.
E) 4.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these square planar complex ions can have cis-trans isomers?
A) [Pt(NH3) 4]2+
B) [Ni(NH3) 4]2+
C) [Pt(NH3) 2Cl2]
D) [Pt(NH3) Cl3]-
E) [Ni(NH3) 3Cl]+
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The electron configuration of a Ti atom is
A) [Ne]3s23d2.
B) [Ne] 3s24d2.
C) [Ar]4s23d2.
D) [Ar]4s24d2.
E) [Ar]3d4.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The best name for the complex shown below is 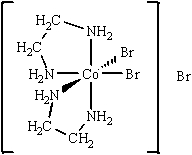
A) cobalt(III) bis(ethylenediamine) bromide.
B) dibromobis(ethylenediamine) cobalt(III) bromide.
C) dibromidedi(ethylenediamine) cobalt(III) bromide.
D) dibromodiethylenediaaminecobalt(III) bromide.
E) tribromobis(ethylenediamine) cobalt(III) .
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these ligands produces the strongest crystal field?
A) Cl-
B) CO
C) OH-
D) H2O
E) NH3
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Predict the number of unpaired electrons in the [Cr(en)3]2+ ion.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the coordination compound [Cr(NH3) (en) 2Cl]Br2, the coordination number (C.N.) and oxidation number (O.N.) of the metal atom are, respectively,
A) C.N. = 6; O.N. = +4.
B) C.N. = 6; O.N. = +3.
C) C.N. = 5; O.N. = +2.
D) C.N. = 4; O.N. = +2.
E) C.N. = 4; O.N. = +3.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The total number of electrons in the 3d orbitals of Co3+ is
A) 4.
B) 5.
C) 6.
D) 7.
E) 10.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these complex ions would absorb light with the longest wavelength?
A) [Co(H2O) 6]2+
B) [Co(NH3) 6]2+
C) [CoF6]4-
D) [Co(CN) 6]4-
E) [Co(en) 6]2+
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Name the complex ion [Cr(en)2(H2O)2]2+.
Correct Answer

verified
diaquobis(...View Answer
Show Answer
Correct Answer
verified
View Answer
True/False
Bidentate and polydentate ligands are also called chelating agents.
B) False
Correct Answer

verified
Correct Answer
verified
Essay
Write the chemical formula of the dibromobis(oxalato)cobaltate(III)ion.
Correct Answer

verified
Correct Answer
verified
Short Answer
Predict the number of unpaired electrons in the [CrCl6]4- ion.
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the oxidation number of cobalt in [Co(NH3)6]Cl3?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In K4[Fe(CN) 6], how many 3d electrons does the iron atom have?
A) 3
B) 4
C) 5
D) 6
E) 7
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The total number of electrons in the 3d orbitals of a titanium atom is
A) 1.
B) 2.
C) 3.
D) 4.
E) 5.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 76 of 76
Related Exams