A) Br2
B) Cl2
C) F2
D) I2
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following data to determine the molar heat of vaporization of chlorine. T (°C) -84.5 -71.2 -47.3 P (mmHg) 40.0 100.0 400.0
A) 34,700 J
B) 21,900 J
C) 317 J
D) 712 J
E) 9.99 kJ
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the amount of heat needed to melt 2.00 kg of iron at its melting point (1,809 K) , given that Hfus = 13.80 kJ/mol.
A) 494 kJ
B) 27,600 kJ
C) 27.6 kJ
D) 27,600 J
E) 25,000 kJ
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
Of the given pair of compounds, which would have the higher boiling point? CCl4 or CH4
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The molar enthalpy of vaporization of hexane (C6H14) is 28.9 kJ/mol, and its normal boiling point is 68.73°C. What is the vapor pressure of hexane at 25°C?
A) 171 torr
B) 4.44 torr
C) 117 torr
D) 3370 torr
E) 759 torr
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following substances should have the lowest boiling point?
A) CBr4
B) CBr3F
C) CBr2F2
D) CBrF3
E) CF4
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Indicate all the types of intermolecular forces of attraction in SF6(g).
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For which of the following species are the dispersion forces strongest?
A) C4H10
B) C5H12
C) C6H14
D) C7H16
E) C8H18
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Identify the dominant (strongest)type of intermolecular force present in RbCl(s).
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following substances crystallizes as a molecular solid?
A) KI
B) SiO2
C) Sn
D) CH3OH
E) Al2(SO4) 3
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Calculate the amount of enthalpy required to heat 25.0 g of solid benzene (C6H6)at -10°C to liquid benzene at 20.0°C. Thermodynamic data for benzene: specific heat of solid benzene = 1.52 J/g·°C; specific heat of liquid benzene = 1.73 J/g·°C; melting point = 5.5°C; Hfus = 9.9 kJ/mol.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The vapor pressure of a liquid in a closed container depends upon
A) the amount of liquid.
B) the surface area of the liquid.
C) the volume of the container.
D) the temperature.
E) None of the above.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following liquids would have the lowest viscosity at 25°C?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following crystallizes in a metallic lattice?
A) C
B) NaMnO4
C) K
D) LiClO4
E) K2Cr2O7
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following in order of increasing boiling point: RbCl, CH3Cl, CH3OH, CH4.
A) CH3OH < CH3Cl < RbCl < CH4
B) CH3OH < CH4 < CH3Cl < RbCl
C) RbCl < CH3Cl < CH3OH < CH4
D) CH4 < CH3OH < CH3Cl < RbCl
E) CH4 < CH3Cl < CH3OH < RbCl
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following substances should exhibit hydrogen bonding in the liquid state?
A) PH3
B) He
C) H2S
D) CH4
E) CH3OH
G) All of the above
Correct Answer

verified
Correct Answer
verified
Essay
Ethanol and dimethyl ether have the same molecular formula, C2H6O. Ethanol boils at 78.4°C. Dimethyl ether boils at -23.7°C. Their structural formulas are, respectively, CH3CH2OH and CH3OCH3. Explain why the boiling point of the ether is so much lower than the boiling point of ethanol.
Correct Answer

verified
Ethanol is a polar molecule th...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Boron nitride, BN3, melts at approximately at 3,000°C under high pressure. This material is almost as hard as diamond. What kind of crystal is this?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based on the phase diagram shown below, how will the melting point of the substance change if the pressure is increased above 1 atm? 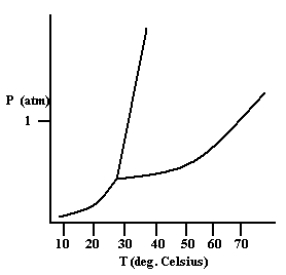
A) The melting point will decrease.
B) The melting point will remain the same.
C) The melting point will increase.
D) The substance will not melt at pressures of 1 atm and above; instead, the solid sublimes to form the gas phase.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of nearest neighbors (atoms that make contact) around each atom in a face-centered cubic lattice of a metal is
A) 2.
B) 4.
C) 6.
D) 8.
E) 12.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 138
Related Exams