A) I only
B) II only
C) III only
D) I and II
E) II and III
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Nitrous oxide, N2O, is sometimes called "laughing gas". What is the formal charge on the central nitrogen atom in the most favorable Lewis structure for nitrous oxide based on minimizing formal charge overall (The atom connectivity is N-N-O.)
A) -2
B) -1
C) 0
D) +1
E) +2
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Lewis dot symbol for the S 2- ion is
A) ![]()
B) ![]()
C) S2-
D) ![]()
E) ![]()
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of resonance structures for the sulfur dioxide molecule that satisfy the octet rule is
A) 1.
B) 2.
C) 3.
D) 4.
E) None of these.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use bond energies to estimate the enthalpy of formation of HBr(g) . BE(H-H) = 436 kJ/mol BE(Br-Br) = 192 kJ/mol BE(H-Br) = 366 kJ/mol
A) +262 kJ/mol
B) +104 kJ/mol
C) +52 kJ/mol
D) -52 kJ/mol
E) -104 kJ/mol
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many covalent bonds will be drawn to bromine in BrO3- for the dot structure that expands the octet to minimize formal charge and if necessary places negative formal charges on the most electronegative atom(s)
A) 3
B) 4
C) 5
D) 6
E) 7
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The total number of valence electrons in the ion SO42- is
A) 24.
B) 26.
C) 28.
D) 30.
E) 32.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following covalent bonds is the most polar (i.e., highest percent ionic character)
A) Al - I
B) Si - I
C) Al - Cl
D) Si - Cl
E) Si - P
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the Lewis structure of the iodate ion, IO3-, that satisfies the octet rule, the formal charge on the central iodine atom is
A) +2.
B) +1.
C) 0.
D) -1.
E) -2.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is most likely to be a covalent compound
A) Rb2O
B) BaO
C) SrO
D) SeO2
E) MnO2
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The following reaction correctly shows the chemical equation for which the enthalpy of reaction is the lattice energy of KCl(s).KCl(s) K+(g) + Cl - (g)
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following solids would have the highest melting point
A) NaI
B) NaF
C) MgO
D) MgCl2
E) KF
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the formal charge on the oxygen atom in N2O (the atomic order is N-N-O)
A) -2
B) -1
C) 0
D) +1
E) +2
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following Lewis structure for OF2 uses how many valence electrons
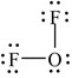
A) 16
B) 18
C) 20
D) zero
E) None of the above
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Lewis structure reveals a triple bond in which of the following molecules
A) Br2
B) O2
C) N2
D) H2
E) CH4
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The total number of bonding electrons in a molecule of formaldehyde (H2CO) is
A) 3.
B) 4.
C) 6.
D) 8.
E) 18.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 121 - 136 of 136
Related Exams