A) CH3(CH2) 2CH3, - 0.5 ℃
B) CH3(CH2) 2CHO, 75 ℃
C) CH3(CH2) 3OH, 100 ℃
D) CH3(CH2) 2COOH, 95 ℃
E) CH3(CH2) 3CHO, 103 ℃
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Carboxylic acids with the general formula CH3(CH2) nCO2H have a nonpolar CH3-CH2- tail and a polar -CO2H head. What effect would increasing the value of "n" have on the solubility of carboxylic acids?
A) The solubility would increase in both water and in nonpolar solvents such as CCl4.
B) The solubility would decrease in both water and in nonpolar solvents such as CCl4.
C) The solubility would increase in water but decrease in CCl4.
D) The solubility would decrease in water but increase in CCl4.
E) The solubility of carboxylic acids in both water and CCl4 would remain the same.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following will be most soluble in a nonpolar solvent such as carbon tetrachloride, CCl4?
A) KI
B) H2O
C) NH3
D) CBr4
E) HF
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rare gases, such as xenon, can be frozen to form solids. The intermolecular forces responsible for maintaining the solid are best described as:
A) hydrogen bonds
B) covalent bonds
C) dipole-dipole forces
D) dipole-induced dipole forces
E) induced dipole-induced dipole forces
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
refer to the phase diagram shown below.
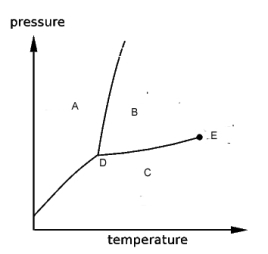 -The regions labeled A,B, and C are
-The regions labeled A,B, and C are
A) the gas, liquid and solid phases, respectively.
B) the solid and liquid phases and the triple point, respectively.
C) the gas and liquid phases and the critical point, respectively.
D) the solid, liquid and gas phases, respectively.
E) pressure, temperature and moles, respectively.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In most cases, reading from left to right across a phase diagram, what is the order in which you would find the three states of matter?
A) gas < liquid < solid
B) gas < solid < liquid
C) liquid < gas < solid
D) liquid < solid < gas
E) solid < liquid < gas
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molality of a bromine solution made by dissolving 39.95 g of Br2 in 5.00 x 102 g of CCl4?
A) 0.0800
B) 0.250
C) 0.500
D) 1.00
E) 2.00
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When a nonvolatile solute is dissolved in a volatile solvent, the vapor pressure, freezing point, and boiling point
A) are all higher for the solution than for the pure solvent.
B) are all lower for the solution than for the pure solvent.
C) change differently, with vapor pressure increasing and boiling and freezing points decreasing.
D) change differently, with boiling point increasing and vapor pressure and freezing point decreasing.
E) do not change because the solute is nonvolatile.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which compound would you expect to have the highest vapor pressure at 25°C?
A) C7H16 (BP = 98.4°C)
B) C8H18 (BP = 125.7°C)
C) C9H20 (BP = 150.8°C)
D) C10H22 (BP = 174.3°C)
E) It is impossible to tell which compound would have the highest vapor pressure because there is no relationship between vapor pressure and the boiling point of a compound.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following characteristics is associated with a low boiling point for a molecular substance?
A) high molecular weight
B) high vapor pressure
C) strong intermolecular forces
D) large dipole moment
E) strong cohesion forces
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which of the following substances do you expect to find the strongest intermolecular hydrogen bonding forces?
A) NH3
B) H2S
C) H2O
D) CF4
E) NaH
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the mole fraction of CCl4 (MW = 154 g/mol) in a solution prepared by dissolving 32 grams of CCl4 in 75 grams of C6H6 (MW = 78.1 g/mol) ?
A) 0.18
B) 0.22
C) 0.30
D) 0.48
E) 0.82
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
Pure benzene melts at 5.5°C. A solution of 1.25 g of CCl4 in 1.00 x 102 g of benzene would have what freezing point? (Benzene: kb = 5.1°C/m)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following concentration units would change when the temperature of the solution changes? (Hint: remember that liquids expand and contract as the temperature changes.) (I) molarity (II) molality (III) mole fraction of solute (IV) mole fraction of solvent
A) I
B) I and II
C) I, II and III
D) all of the above
E) none of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
refer to the table below:
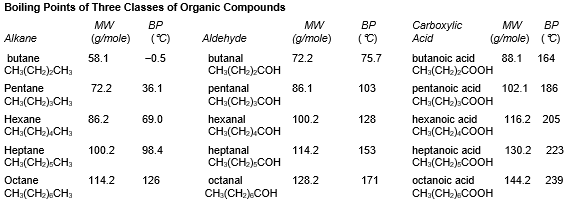 -What intermolecular force is most responsible for the difference in boiling point between octane and pentane?
-What intermolecular force is most responsible for the difference in boiling point between octane and pentane?
A) dispersion
B) dipole-dipole interaction
C) dipole-induced dipole interaction
D) hydrogen bonding
E) all of these
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is true?
A) Molarity (M) is calculated by dividing the number of moles of solute by the number of liters of solvent.
B) Molality (m) is calculated by dividing the number of moles of solute by the number of liters of solvent.
C) Mole fraction is calculated by dividing the number of moles of solvent by the number of moles of solute.
D) Mole fraction is calculated by dividing the number of moles of solute by the number of moles of solvent.
E) Statements (a) through (d) are all false.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which force must be overcome to melt solid pentane, C5H12?
A) metallic bonding
B) covalent bonding
C) ionic bonding
D) dispersion forces
E) all of these must be overcome to melt pentane
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is correct for the solubilities of alcohols with the formula CH3(CH2) nOH?
A) The solubility in water is minimal because the compound is organic.
B) The solubility in water increases as n increases.
C) The solubility in water decreases as n increases.
D) The solubility in water is independent of n.
E) The solubility in water depends only on the extent of hydrogen bonding.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds has the lowest vapor pressure?
A) CCl4
B) CBr4
C) CF4
D) CH4
E) CI4
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following three compounds have similar molecular weights. Place them in order of increasing boiling point. Formaldehyde: H2C=O methanol: CH3OH ethane: CH3CH3
A) formaldehyde < ethane < methanol
B) ethane < methanol < formaldehyde
C) ethane < formaldehyde < methanol
D) methanol < formaldehyde < ethane
E) none of these
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 54
Related Exams