Filters
Question type
A) 7.0 × 10 −6
B) 1.3 × 10 −3
C) 2.2 × 10 4
D) 1.4 × 10 5
E) > 2.0 × 10 5
F) All of the above
G) B) and E)
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Question 52
Multiple Choice
Calculate ΔG° for the reaction SiCl4(g) + 2Mg(s) → 2MgCl2(s) + Si(s)
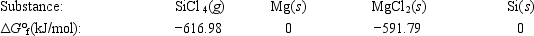
A) 566.60 kJ
B) 50.38 kJ
C) 25.19 kJ
D) −25.19 kJ
E) −566.60 kJ
F) C) and D)
G) B) and C)
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Question 53
Multiple Choice
Which one of the following changes of state increases the entropy of the system?
A) Condensation
B) Cooling a gas
C) Freezing
D) Crystallization
E) Sublimation
F) A) and D)
G) B) and E)
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Question 54
Multiple Choice
You are given pure samples of pentane, CH3CH2CH2CH2CH3(l) , and 1,3-pentadiene, CH2=CHCH=CHCH3(l) . What prediction would you make concerning their standard molar entropies at 298 K?
A) S° pentane > S° 1, 3-pentadiene
B) S�° pentane < S° 1, 3-pentadiene
C) S° pentane ≈ S° 1, 3-pentadiene
D) S° pentane = S° 1, 3-pentadiene + 2 S°H 2
E) More information is needed to make reasonable predictions.
F) C) and D)
G) A) and E)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Question 55
Multiple Choice
Which of the following is always true for an endothermic process?
A) q sys > 0, Δ S surr < 0
B) q sys < 0, Δ S surr > 0
C) q sys < 0, Δ S surr < 0
D) q sys > 0, Δ S surr > 0
E) w < 0
F) C) and D)
G) A) and B)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 85 of 85
Related Exams