A) 0.352 kg
B) 0.122 kg
C) 0.820 kg
D) 0.280 kg
E) 0.632 kg
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements is classified as a semimetal?
A) calcium
B) boron
C) fluorine
D) uranium
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What element is defined by the following information? p+ = 17 n° = 20 e- = 17
A) calcium
B) rubidium
C) chlorine
D) neon
E) oxygen
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following elements is a poor conductor of heat and electricity?
A) copper
B) fluorine
C) iron
D) lead
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Match the following. -magnesium
A) Mg
B) Fe
C) Si
D) K
E) C
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following contains the most atoms? You shouldn't need to do a calculation here.
A) 10.0 g Ne
B) 10.0 g He
C) 10.0 g Ar
D) 10.0 g Kr
E) 10.0 g Mg
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When forming an ion, fluorine, F, will ________ electron(s) .
A) gain one
B) gain two
C) gain three
D) lose one
E) lose two
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Bromine has two naturally occurring isotopes, Br-79 and Br-81. The atomic mass of Br-79 is 78.9183 u with a percent abundance of 50.69% and the atomic mass of Br-81 is 80.9163 u. Calculate the atomic mass of bromine.
A) 80.91 u
B) 78.92 u
C) 79.90 u
D) 80.92 u
E) 80.03 u
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following contains the fewest atoms? You shouldn't need to do a calculation here.
A) 4.0 g Li
B) 4.0 g Na
C) 4.0 g Rb
D) 4.0 g K
E) 4.0 g Ca
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a transition element?
A) Pd
B) Sn
C) K
D) U
E) Pr
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many electrons does the Ca2+ ion possess?
A) 27
B) 18
C) 9
D) 3
E) 20
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the charges of protons, neutrons, and electrons.
A) protons +1, neutrons 0, electrons -1
B) protons 0, neutrons -1, electrons +1
C) protons -1, neutrons 0, electrons +1
D) protons 0, neutrons +1, electrons -1
E) protons +1, neutrons -1, electrons 0
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the charge that a calcium ion would have.
A) 6-
B) 2-
C) 3+
D) 2+
E) 1+
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many protons (p) and neutrons (n) are in an atom of 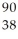 Sr?
Sr?
A) 38 p, 52 n
B) 38 p, 90 n
C) 52 p, 38 n
D) 90 p, 38 n
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the chemical symbol of element Q in 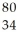 Q.
Q.
A) Br
B) Hg
C) Pd
D) Se
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Match the following. -carbon
A) Mg
B) Fe
C) Si
D) K
E) C
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many neutrons are in arsenic-42?
A) 33
B) 41
C) 9
D) 41.9
E) 75
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the atomic mass of chromium if chromium has four naturally occurring isotopes with the following masses and natural abundances: Cr-50 49.9461 u 4.35% Cr-52 51.9405 u 83.79% Cr-53 52.9407 u 9.50% Cr-54 53.9389 u 2.36%
A) 51.94 u
B) 51.69 u
C) 208.7 u
D) 53.21 u
E) 52.00 u
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is FALSE according to John Dalton's reformulated atomic theory?
A) Each element is composed of destructible particles called atoms.
B) All atoms of a given element have the same mass as well as other properties.
C) Compounds are formed when atoms combine in simple, whole-number ratios.
D) Atoms composing one element cannot change into atoms of another element during a chemical reaction.
E) All atoms of a given element have the same physical and chemical properties.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Isotopes differ in the number of ________.
A) protons
B) neutrons
C) electrons
D) neutrons and electrons
E) protons and electrons
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 174
Related Exams